 In other words, Moseley had found that elements are different from one another because their atoms have different numbers of protons. When he looked at the frequencies emitted by a series of elements, he found a pattern that was best explained if the positive charge in the nucleus increased by exactly one unit from element to element. Moseley found that each element he studied emitted x-rays at a unique frequency. The explanation came in 1913 from Henry Moseley, who fired electrons at atoms, resulting in the emission of x-rays. In fact, it took 44 years for the correct explanation of the regular patterns in Mendeleev's periodic table to be found. In 1869 the electron itself had not been discovered - that happened 27 years later, in 1896. Of course, this was perfectly reasonable when we consider scientific knowledge in 1869. Mendeleev believed, incorrectly, that chemical properties were determined by atomic weight. With the benefit of hindsight, we know that Mendeleev's periodic table was underpinned by false reasoning. Similarly, Mendeleev discovered gallium ( eka-aluminum) and scandium ( eka-boron) on paper, because he predicted their existence and their properties before their actual discoveries.Īlthough Mendeleev had made a crucial breakthrough, he made little further progress. He called this new element eka-silicon, after observing a gap in the periodic table between silicon and tin: Just as Adams and Le Verrier could be said to have discovered the planet Neptune on paper, Mendeleev could be said to have discovered germanium on paper. Mendeleev realized that the table in front of him lay at the very heart of chemistry.Īnd more than that, Mendeleev saw that his table was incomplete - there were spaces where elements should be, but no-one had discovered them. For example, magnesium (atomic weight 24.3) is placed to the right of sodium (atomic weight 23.0): A higher atomic weight than the one on its left. Mendeleev found he could arrange the 65 elements then known in a grid or table so that each element had:ġ. The periodic table we use today is based on the one devised and published by Dmitri Mendeleev in 1869. It bonds to carbon covalently in carbon tetrachloride. Many elements can form covalent bonds depending on the circumstances.įor example, the bonding in water, H 20, is mainly covalent Īnd while chlorine is bonded to sodium ionically in sodium chloride, In fact, if they are going to form ions, Group 14 elements form positive ions.Ĭarbon's millions of organic compounds are all based on shared electrons in covalent bonds. It obtains a noble gas structure by sharing electrons with other atoms.Įlements in Groups 15,16 and 17, find it easier to gain electrons than lose them.įor example, oxygen atoms gain two electrons to form O 2- ions.Įlements in Group 14 could lose four, or gain four electrons to achieve a noble gas structure. Most elements in Group 3 lose three electrons to form 3+ ions.īoron, however, shows little tendency to form ions. 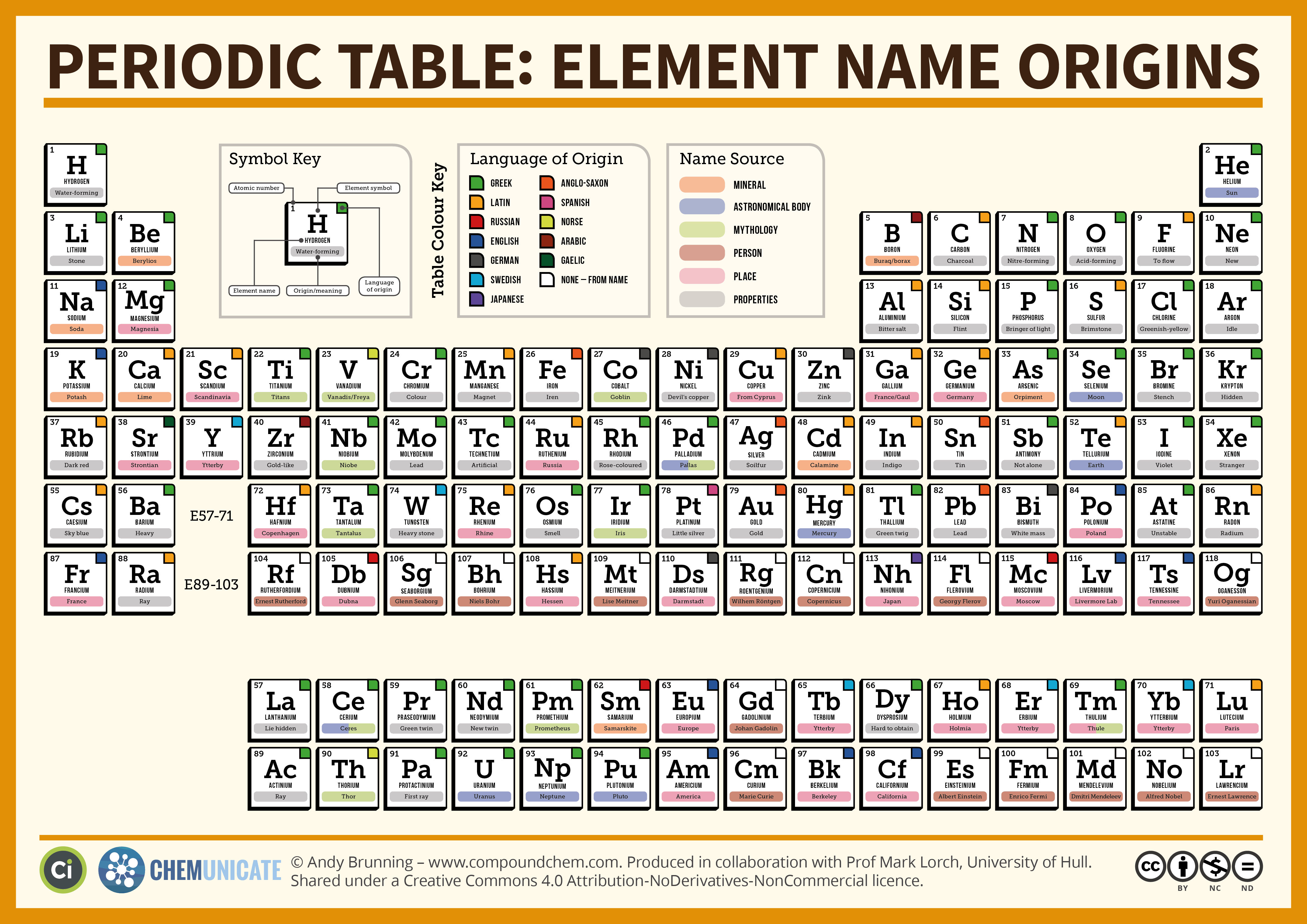
These have the same electron configuration as the noble gas neon. Group 2 atoms lose two electrons to form positively charged ions.įor example, magnesium atoms form Mg 2+ ions. These ions have the same stable electron configuration as the noble gas helium.Īll Group 1 atoms can lose one electron to form positively charged ions.įor example, potassium atoms do this to form ions with the same electron Li atoms readily give up one electron to form positively charged, Li + ions. The outer shell is incomplete, and therefore unstable. Be sure to memorize silver (Ag + ) and zinc ( Zn 2 + ) charges in addition to this chart.A lithium atom has two electrons orbiting in an inner shell and only one electron orbiting in its outer shell. 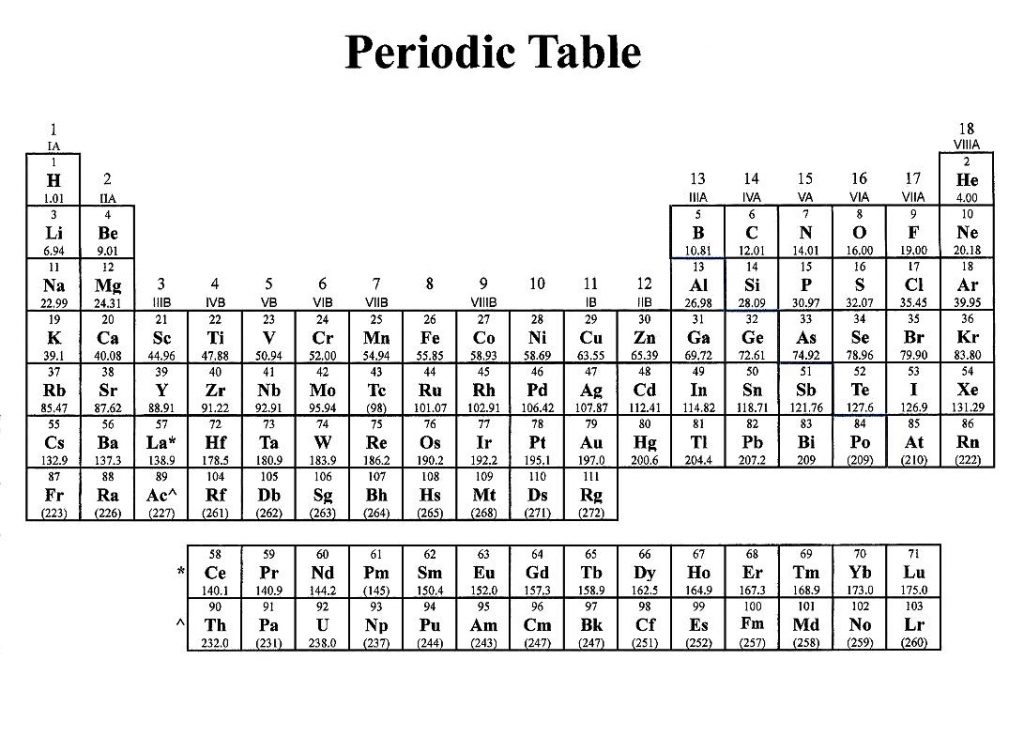
Within a group (family) of elements, atoms form ions of a certain charge. 
The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
